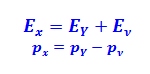MODERN PHYSICS – 2020
Recent entrance exams
01- (ENEM-MEC-019)

When one considers the extreme speed with which light spreads in all directions and that, when it comes from different places, even totally opposite, [the light rays] pass through each other without hindering each other, one understands that, when we see a luminous object, this could not occur by the transport of matter that comes from the object to us, as an arrow or bullet passes through the air; for this certainly is quite repugnant to these two properties of light, especially the last.

HUYGENS, C. In: MARTINS, RA Treatise on Light, by Christian Huygens. History and Philosophy of Science Notebook, suppl. 4, 1986.
The text challenges which conception about the behavior of light?
(A) The understanding that light needs a propagation medium, spread by defenders of the existence of ether.
(B) The wave model for light, which considers the possibility of interference between light beams.
(C) The corpuscular model defended by Newton, which describes light as a beam of particles.
(D) The belief in the infinite speed of light, defended by most Greek philosophers.
(E) The idea defended by the Greeks that light was produced by the eyes
Particle is basically composed of matter even if the mass is almost negligible.
A wave is a periodic disturbance, that is, regular, that transports only energy and not matter.
This part of the statement “ …. light spreads everywhere and that, when they come from different places, even totally opposite, [the light rays] pass through each other without getting in each other’s way, it is understood that, when we see a luminous object” refers to the wave theory of light, (theory of the independence of light rays).
The last text he refers to “..this could not occur by the transport of matter that comes from the object to us, like an arrow or bullet passes through the air” he is contesting the transport of matter (arrow or bullet) on the corpuscular model (light as a beam of particles) proposed by Newton which is not in agreement with the wave model proposed by HUYGENS.
R-C
02- (São Paulo State College of Technology – FATEC – 2020)

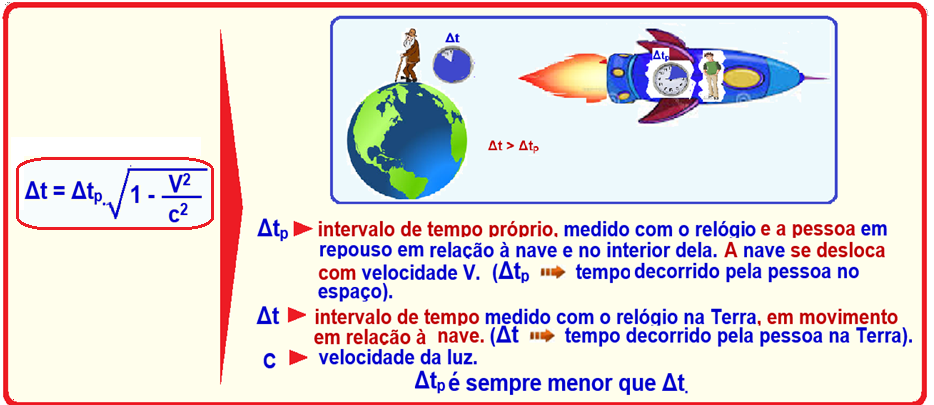
In Einstein’s Special Theory of Relativity, two concepts studied refer to the fact that,

when considering an object propagating at the speed of light, we can verify
(A) time dilation and length dilation.
(B) the contraction of time and the dilation of length.
(C) time dilation and length contraction.
(D) time dilation without length contraction.
(E) time contraction without length contraction
Time dilation
Variation of space (length)
R-C
03- (FGV – SP – Economy – 2020)


Rule of three:

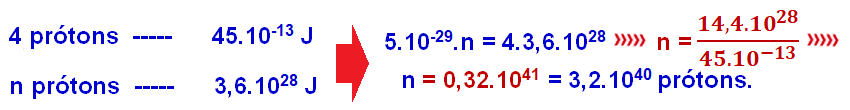
R-C
04- (Faculty of Medical Sciences of the Santa Casa of São Paulo – SP – 2020)

![]()



R- And
05- (UDESC – Santa Catarina State University – 2020)

Analyze the propositions in relation to the concepts of Modern Physics.

I. The speed of light, in a vacuum, is the same for any inertial system and depends on the motion of the light source.
II. Light, for absorption and emission phenomena, has a wave character.
III. The phenomenon of electron diffraction shows that corpuscular particles can also have wave-like behavior.
IV. One of the main fundamental aspects of quantum mechanics is that energy in nature can assume discrete values.
V. One of the fundamental principles of relativity is that only massless particles can reach the speed of light.
Select the correct alternative .
A. ( ) Only statements I, II and III are true.
B. ( ) Only statements III, IV and V are true.
C. ( ) Only statements I, II and IV are true.
D. ( ) Only statements I, II and V are true.
E. ( ) Only statements II, IV and V are true.
I. False
The speed of light, in a vacuum, is the same for any inertial system and is independent of the
movement of the light source.
II. False
The intercalation of ideas between waves and particles in relation to light is accepted in the scientific community as the Dual Nature of Light; because in certain phenomena (interference, refraction, diffraction…) the electromagnetic theory can explain and the corpuscular theory is associated with the phenomena of absorption and emission of energy.
III. Correct
IV. Correct
Quantum mechanics ( also known as quantum physics , quantum theory , wave mechanics model , and matrix mechanics ) is the physical theory that succeeds in studying physical systems whose dimensions are close to or below the atomic scale, such as molecules, atoms, electrons,
protons and other subatomic particles, although it can also describe macroscopic phenomena in many cases.
V. Correct
According to the theory of relativity, particles with mass at speeds close to that of light would have their masses tending towards infinity.
R-B
06- (UEG – GO – MEDICINE – 2020)

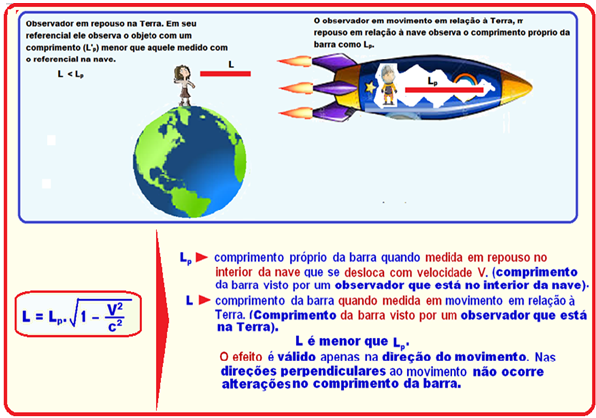
A space station monitors the speed of a spacecraft by measuring its length and comparing it to when the spacecraft is parked on its launch pad.

At a certain point, the station found that the length of the ship had reduced by 29% compared to that measured at rest.
To the space station this ship was traveling, in terms of the speed of light, approximately
a) 0.9c
b) 0.7c
c) 0.2c
d) 0.5c
e) 0.3c
Pay close attention to the form below
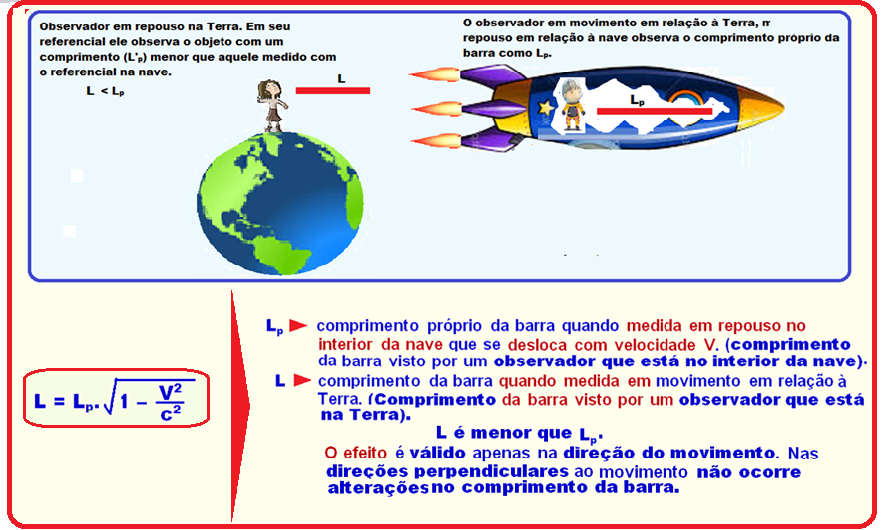

R-B
07- ( Federal University of Lavras – UFLA – MG – 2020)

When the speeds of objects are close to the speed of light, the following postulates are considered: the speed of light is constant, and is the same for all observers,

regardless of their state of motion, and the laws of physics are the same for all observers.
Based on these statements, analyze the case of two spaceships traveling in the same direction, but with opposite directions, and that intermittently fire beams of light inside

of the ships. If each ship has speed v and the speed of light is symbolized as c, indicate the speed of the light beams from one of the ships perceived by the pilot of the other ship .
(A) Speed c
(B) Speed vc
(C) Speed v+c
(D) Speed v

A- A
08- Federal University of Uberlândia – UFU – MG – mid-year – 2019/2020

There are processes that occur in the electronic structure of atoms in which an electron can gain or lose energy. In these processes , the electron passes from one energy level to another, and the energy difference between these two levels, in some of these processes, can be emitted as a photon of light.
The photon has energy that can be determined by a direct relationship with the frequency of light through the equation E = h . f, where E is the energy of the photon, h is Planck’s constant (h = 6.6 x 10 -34 Js) and f is the frequency of the emitted light. In these situations, a widely used unit of energy is the electronvolt (eV), where 1 eV = 1.6 x 10 -19 J.
Consider two electronic energy levels with values of E 1 = -2.93 eV and E 2 = -1.28 eV, and an electron that decays from the E 2 level to the E 1 level , emitting a photon.
What is approximately the frequency of light associated with this photon?
A) 4.00 x 101 4 Hz
B) 2.42 x 10 15 Hz
C) 1.00 x 10 15 Hz
D) 6.64 x 10 13 Hz
Bohr atomic model
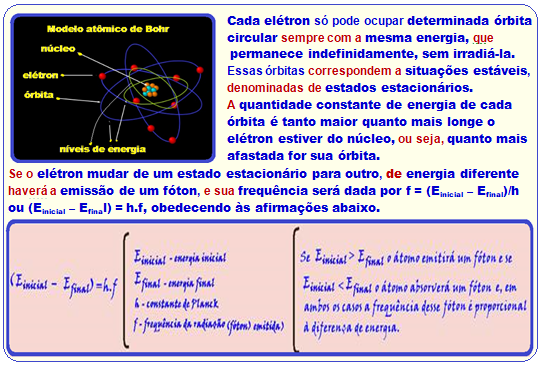
Calculation of the energy (W) contained in a photon

E = E 2 – E 1 = – 1.28 eV – (- 2.93 eV) = 1.65 eVx1.6.10 -19 = 2.64.10 -19 J; h = 6,6.10 -34 Js and f = ?
E = hf ![]() 2,64.10 -19 = 6,6.10 -34 .f
2,64.10 -19 = 6,6.10 -34 .f ![]() f = 2,64.10 -19 /6,6.10 -34 = 0,4.10 15 f = 4 . 10 14 Hz.
f = 2,64.10 -19 /6,6.10 -34 = 0,4.10 15 f = 4 . 10 14 Hz. ![]()
A- A
09- (UNESP – SP – 2020)

The visual sensitivity of humans and animals lies within a narrow range of the electromagnetic radiation spectrum, with wavelengths between 380 nm and 760 nm.
It is remarkable that plants also react to radiation within this same range, including photosynthesis and phototropic growth.

The reason for the importance of this narrow band of electromagnetic radiation is that the energy carried by a photon is inversely proportional to the wavelength.
Thus, the longer wavelengths do not carry enough energy in each photon to produce an appreciable photochemical effect, and the shorter ones carry energy in an amount that damages organic materials.
(Knut Schmidt-Nielsen. Animal physiology: adaptation and environment , 2002. Adapted.)
The table shows the wavelength of some colors in the visible light spectrum:
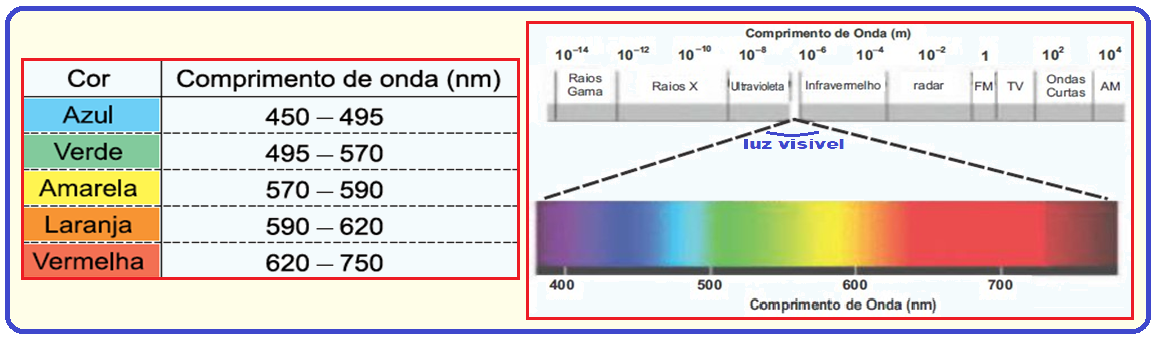

(A) blue.
(B) green.
(C) yellow.
(D) orange.
(E) red.

Fundamental wave equation


R-B
10- Regional University of Cariri (URCA)- CE – 2020

Regarding the nature of Light, mark the alternative far from the scientific context:

A) The physicist Christian Huygens, in 1678, considered Light as consisting of waves and not of
corpuscle as defended by physicist Isaac Newton;
B) Regarding the wave theory, both the corpuscular theory and the color phenomena are explained;
C) The phenomenon of interference is a typically wave-like process not presented
considering light as a corpus entity D) Light is related to electromagnetic waves emitted by the movement of electric charges that
generate electric fields that vary and create magnetic fields;
E) Electromagnetic waves with lengths ranging from meters to kilometers are called visible light.
The false alternative is E , since the wavelengths of visible light are classified according to color, from violet, which has a wavelength λ = 4. 10 -7 m, to red, whose wavelength is λ = 7. 10 -7 m.
R- And
11- (Aeronautics Technical Institute – ITA – SP – 2020)

Cosmic rays interact with atoms in the atmosphere and produce unstable particles X. Through experiments, it is found that X decays into a particle Y and a neutrino ν, according to the decay equation X→Y+ν.

Considering the rest mass of the neutrino to be negligible and X initially at rest, determine the velocity of particle Y in terms of c and the masses of X and Y.
Source: Own authorship

This problem involves conservation of energy and momentum, the difference being that particles travel at very high speeds, making it necessary to use conservation laws in the context of special relativity. Previously, there was particle X with energy E X and momentum P X , then the particle separated and the energies E Y and E ν for each particle came into play, as well as momenta P Y and P ν . Assuming opposite directions for each particle, we have:
Since the statement asked us to disregard the mass of ν , we must use the following approximation for the formula for the energy of this particle :