Latent Heat – Heat exchange with change of state – EN
Latent Heat – Heat exchange with change of state
Latent Heat
The heat received by a body can cause its temperature to increase or for it to undergo a change of state . In the second case you have a quantity of latent heat , that is, the heat
latent refers to the amount of heat a body receives just to change state, which occurs at constant temperature.
It is found that the amount of latent heat (Q) given or removed from a substance so that it changes state, which always occurs at a constant temperature , is directly proportional to its mass (m), and depends on the characteristics of the substance (L). Mathematically:

Meaning of Latent Heat
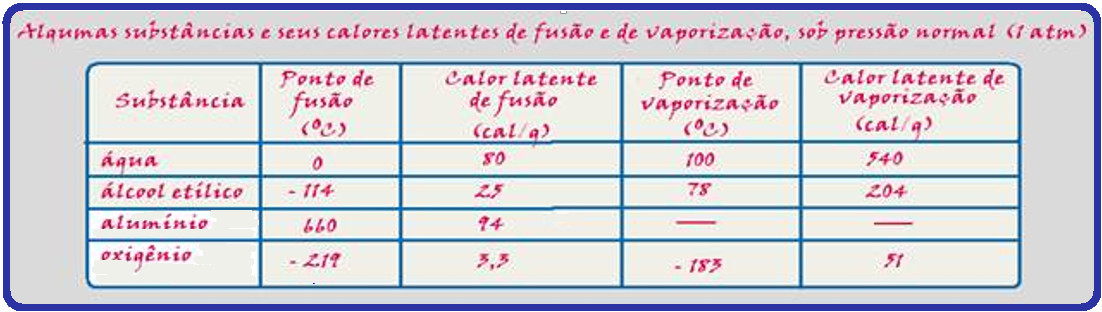
If you want to know, for example, how many calories it takes to melt 200g of aluminum , you should get ![]() Q = mL = 200.94
Q = mL = 200.94 ![]() Q = 18,800 cal or Q = 18.8 kcal.
Q = 18,800 cal or Q = 18.8 kcal.
Heating curve – sensible heat and latent heat
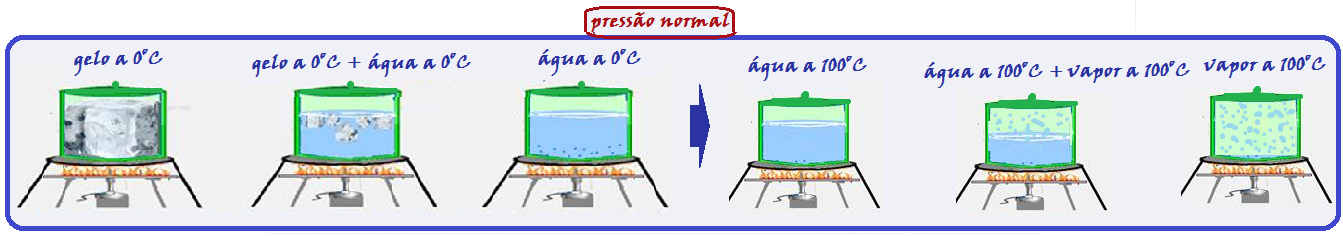
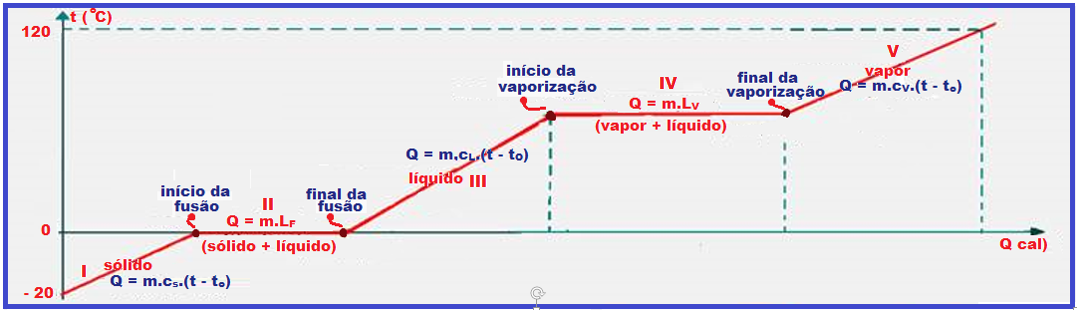
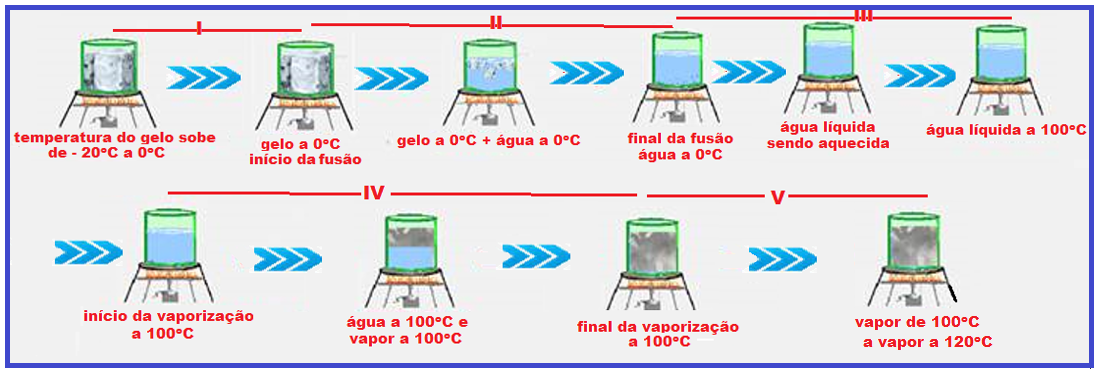
Look very carefully at the graph below and the sequence of figures that illustrate the heating, for example, of a certain mass of water being heated from ice at -20 o C to water vapor at 120 o C.
What you should know, information and tips
![]()
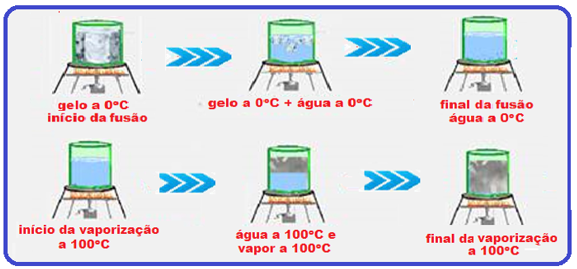
During melting and vaporization the temperature remains constant (0 ° C in melting and 100 ° C in vaporization).
vaporization) because the thermal energy (heat) supplied to the ice was used only to convert it into liquid water and the thermal energy (heat) supplied to the liquid water was used only to convert it into vapor , causing the temperature in these two changes of physical state to remain constant (0 o C in melting and 100 o C in vaporization).
![]()
When you state that the latent heat of fusion of ice is 80 cal/g , you mean that for the melting of 1g of ice or for the solidification of 1g of water, you must give up or take away 80 cal.
![]()
Heating curve – sensible heat and latent heat

![]()
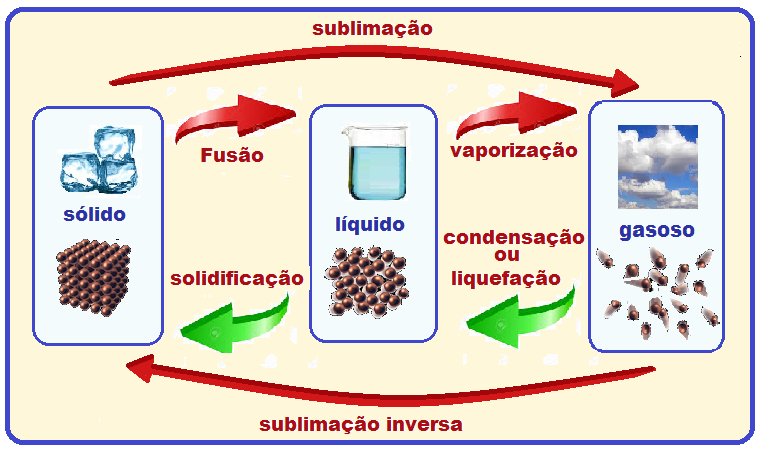
Changes in physical state
![]()
Solving some exercises on heat exchanges involving sensible (specific) heat and latent heat:

Data: specific heat of ice = 0.5 cal/g°C ; specific heat of water = 1 cal/g°C and latent heat of fusion of ice = 80 cal/g
Resolution:
From ice at -20 o C to ice at 0 o C ![]() there is no change of state
there is no change of state ![]() sensible heat
sensible heat ![]() Q 1 = mc ice .
Q 1 = mc ice .
∆ θ = 2.0.5.[0 – (-20)] ![]() Q 1 = 20kcal (heat used to heat the ice).
Q 1 = 20kcal (heat used to heat the ice).
From ice at 0 o C to water at 0 o C ![]() is a change of state (fusion) at constant temperature
is a change of state (fusion) at constant temperature ![]() latent heat of fusion
latent heat of fusion ![]() Q 2 = mL = 2.80 Q 2 = 160 kcal (heat used only in the change of state).
Q 2 = mL = 2.80 Q 2 = 160 kcal (heat used only in the change of state). ![]()
From water at 0 o C to water at 50 o C ![]() there is no change of state
there is no change of state ![]() sensible heat
sensible heat ![]() Q 3 = mc water . ∆θ = 2.1.(50 – 0) Q 3 = 100 kcal (heat used to heat the water to the required temperature, 50 o C).
Q 3 = mc water . ∆θ = 2.1.(50 – 0) Q 3 = 100 kcal (heat used to heat the water to the required temperature, 50 o C). ![]()
The total amount of heat in this heating was Q total = 20 + 160 + 100 = 280 kcal.
As the thermal source supplies 20 kcal per minute ![]() Δt =
Δt = 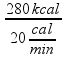 = 14minx60 = 840 s.
= 14minx60 = 840 s.

At a picnic, in order to obtain ice-cold water, it was mixed in a thermos bottle ,
negligible thermal capacity, 2 kg of crushed ice at 0 ° C and 3 kg of water that were in bottles outdoors, at an ambient temperature of 40 ° C.
Disregarding heat exchange with the external environment and knowing the latent heat of fusion of ice (80 cal/g) and the specific heat of water (1 cal/g°C).
Calculate the mass of ice water available for drinking, in kg, after thermal equilibrium has been established.
Resolution:
Heat required to melt 2 kg of ice at 0 °C ![]() Q 1 = mL = 2000.80 = 160000 cal (heat used to melt 2 kg of ice).
Q 1 = mL = 2000.80 = 160000 cal (heat used to melt 2 kg of ice).
Heat required to cool the 3 kg of water from 40 ° C to 0 °C Q = mcΔθ = 3000.1.(0-40) = ![]()
– 120000 cal (heat removed from 3 kg of to go from 40 o C to 0 o C.
Note that the heat released by the water, 120 kcal , is not enough to melt all the ice (since 160 kcal are needed ), ![]() therefore thermal equilibrium will occur at 0 °C , with the 3 kg of original liquid water remaining liquid and a portion of the ice melting worth
therefore thermal equilibrium will occur at 0 °C , with the 3 kg of original liquid water remaining liquid and a portion of the ice melting worth ![]() Q = mL 120000 = m.80 m = = 1500 g = 1.5 kg.
Q = mL 120000 = m.80 m = = 1500 g = 1.5 kg. ![]()
![]()
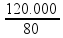
Thus, the final mass of ice water will be 3 kg (liquid already existing at 0 ° C) + 1.5 kg (ice that melted ) = 4.5 kg.
